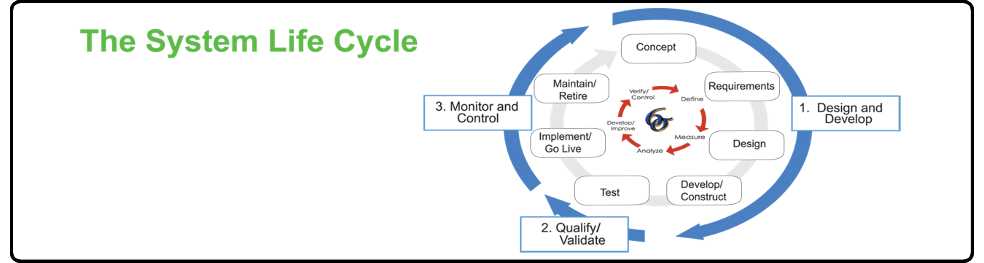
With expertise in all aspects of the system life-cycle, Dedicare can help you develop and validate your manufacturing systems including Facilities, Utilities, Equipment, Processes and Test Methods.
PROCESS DEVELOPMENT
Dedicare is familiar with the latest tools and techniques for process development and improvement including: DOE, Six Sigma, Lean and Risk Management (FMEA). Dedicare provides Process Engineering Management services rooted in 17 years of hands-on process development, improvement, and leadership. Tap into the manufacturing experience from a diverse group of products and manufacturing technologies.
Products:
| Atherectomy Devices |
Catheters |
Guidewires |
| SS and Nitinol Stents |
Drug Eluting Stents |
Orthopedic Implants |
| Glucose Monitoring |
Angioplasty |
Laparoscopy |
Manufacturing Technologies:
| Manual Assembly |
Stent Crimping |
Centerless Grinding |
CNC Machining |
| Welding (laser, plasma, RF) |
Laser Cutting |
Electroploshishing |
Microblasting |
| Honing |
Coiling |
Swaging |
Vapor Deposition |
| Spray Coating |
Extrusion |
Molding |
Drying |
| Tumbling |
Packaging |
Filling |
Formulation |
| Label Printing |
Pressing and Forming |
Polymer Coating |
Cleaning |
TEST METHOD DEVELOPMENT
Dedicare can help you select the appropriate test method, validate it and implement it. Dedicare is familiar with the following measuring equipment and methods:
| Pull Testing |
Vision Systems |
Coordinate Measuring Systems |
Visual Inspection |
| Go/No Go Gaging |
Calipers |
Indicators |
Gage Blocks |
| Micrometers |
Pin and Ring Gaging |
Comparators |
...and more |
SYSTEM VALIDATION
Recognized by peers as an expert in all aspects of systems validation (IQ, OQ, PQ, PPQ), Dedicare provides full service including the development of the governing quality procedures, assessment, validation master planning, and remediation. In addition to process validation, Dedicare also has expertise with Utilities and Facilities qualification (FAT, SAT, Commissioning), Test Method Validation, Cleaning Validation and Software Validation.